
REC – Submission
World class services
for your research
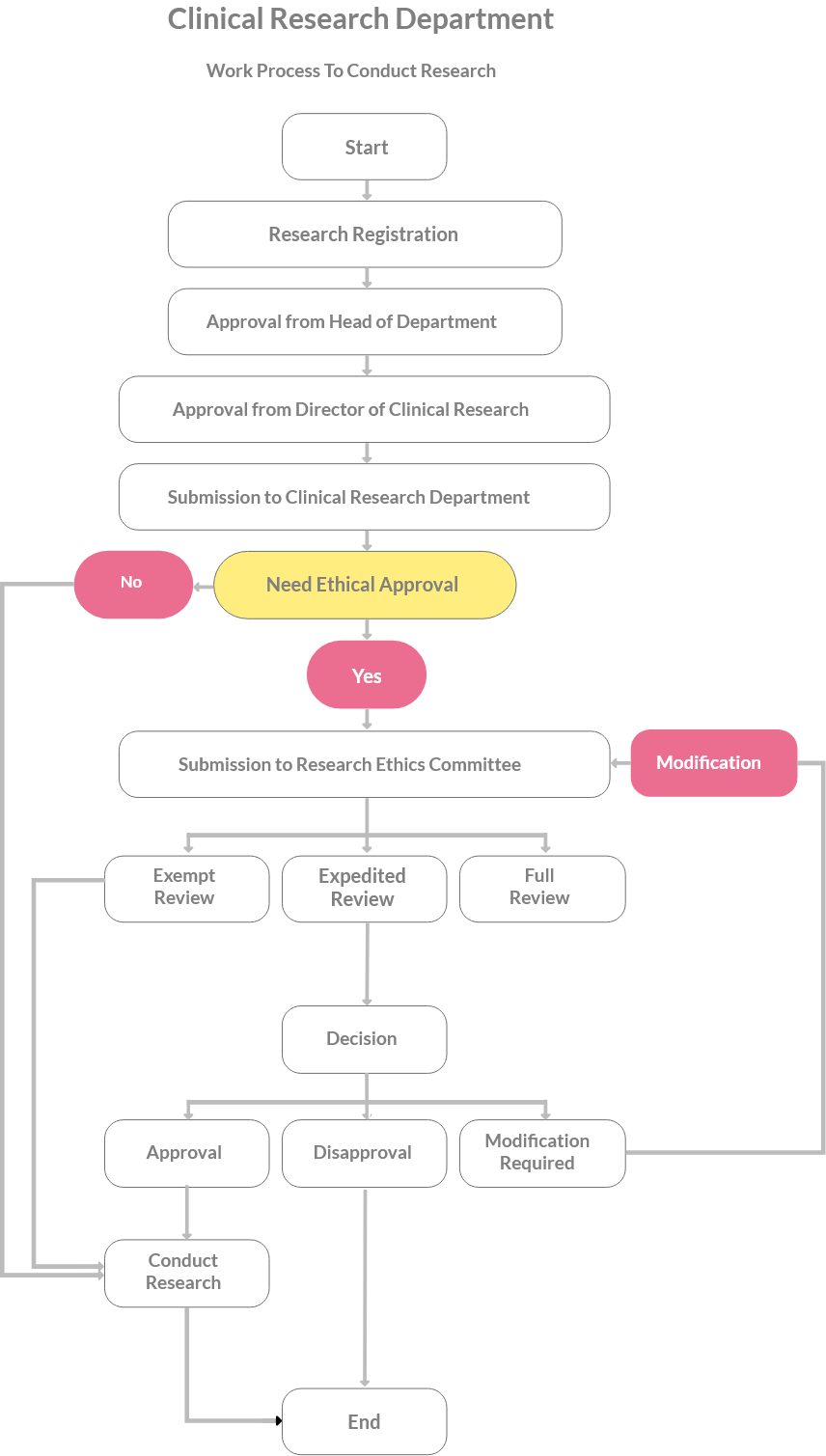
Documents Required
Research and development Project
Registration Form (CRD-QR-AD1)
Documents Required
CRD Determination of Human Subject Research Checklist (CRD-QR-A13)
Documents Required
IJNREC Application Form (CRD-QR-A02) IJNREC Submission Checklist (CRD-QR-A04)
Documents Required
IJNREC Determination Form (CRD-QR-A05)
IJNREC Review Checklist (CRD-QR-A06)
Documents Required
IJNREC Application for Amendment Form (CRD-QR-A07)
IJNREC SAE Reporting Form (CRD-QR-A08)
IJNREC Notification of Protocol Deviation/ Violation Form (CRD-QR-A09)
IJNREC for Renewal Form (CRD-QR-A10)
IJNREC Notification of Closure Form (CRD-QR-A32)
Applicant
Research Ethics Committee
